Characterizing the Effect of the Extravascular Environment on Trypanosoma brucei Antigenic Diversity
Summer 2024 — Mugnier Lab, Johns Hopkins Bloomberg School of Public Health
Funded by the Basic Science Institute Summer Internship Program (BSI-SIP), Johns Hopkins School of Medicine.
Abstract
Trypanosoma brucei poses a significant public health and economic burden across sub-Saharan Africa. This single-celled parasite relies on antigenic variation — periodically switching its dense variant surface glycoprotein (VSG) coat — to evade host antibodies and establish chronic infection. While most research has focused on bloodstream-stage parasites, recent evidence suggests that the primary reservoirs of antigenic diversity reside in extravascular spaces, the fluid-filled gaps between tissue cells. Understanding what triggers VSG switching in these microenvironments is critical for elucidating the parasite’s immune evasion strategy.
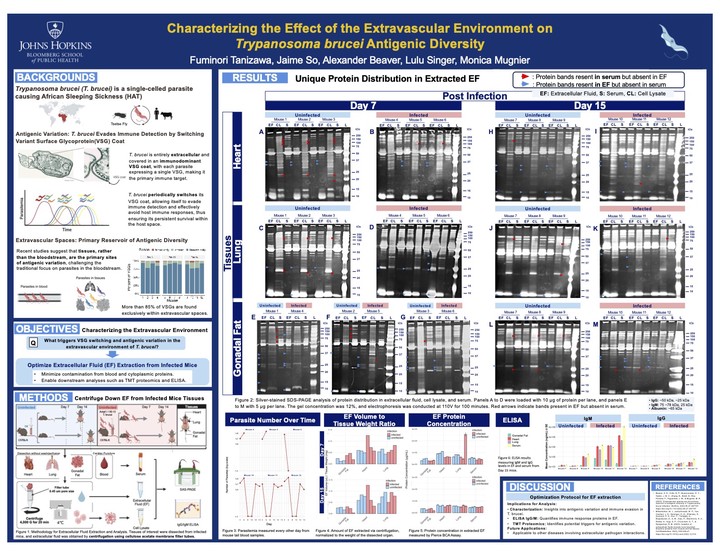
In this project, I optimized a protocol for extracting extracellular fluid (EF) from infected mouse tissues for downstream analyses including TMT proteomics and ELISA. I dissected adipose tissue, heart, and lung, then tested centrifugation speeds, washing, and perfusion techniques to maximize EF yield while minimizing cytoplasmic contamination from tissue damage. SDS-PAGE confirmed that the resulting EF contained a protein profile distinct from both cell lysate and serum. These findings provide a reproducible and standardized method for characterizing the extravascular environment, laying the groundwork for future studies of T. brucei immune evasion and persistence in host tissues.