HAT Complex Protein EAF6: Role in Lifecycle Differentiation of Trypanosoma brucei
Academic Year 2023–2024 — Schulz Lab, Harvey Mudd College
Abstract
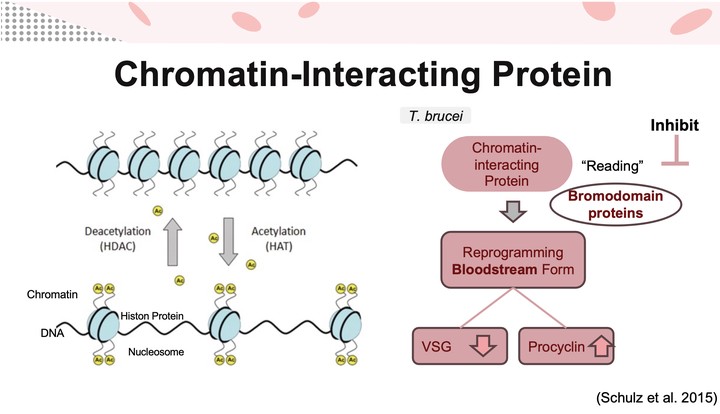
At the Schulz Lab, I investigated the role of EAF6 — a non-catalytic subunit of the histone acetyltransferase (HAT) complex — in the lifecycle differentiation of Trypanosoma brucei, the causative agent of African trypanosomiasis (sleeping sickness). T. brucei evades the host immune system in its bloodstream form by expressing variant surface glycoproteins (VSG), while the insect-stage procyclic form expresses procyclin. The transcriptional mechanisms governing this developmental switch remain incompletely understood.
To determine whether EAF6 is required for differentiation, I designed and cloned RNAi vectors to knock down EAF6 expression, then monitored differentiation in both bloodstream and insect-stage environments by tracking EP1-GFP expression over three days via flow cytometry. EAF6 knockdown cells (+Dox) showed no significant difference in differentiation rate compared to controls (−Dox), though both differentiated more slowly than wild-type parasites. These results suggest that our RNAi clone may have had regulatory issues rather than reflecting a true null phenotype.
Future directions include repeating experiments with independently transformed clones, Western blot validation of EAF6 knockdown, and investigating other HAT complex components such as BDF6. This work contributes to our understanding of chromatin-regulatory proteins as potential therapeutic targets in African trypanosomiasis.